Recent News

Jun Oike, MD, PhD.
Excellent Oral Presentation Award Session at the Japanese Orthopaedic Association (JOA) 2026 Annual Meeting
Congratulations to Jun Oike, MD, PhD on having his abstract selected for the Excellent Oral Presentation Award Session at the 2026 JOA Annual Meeting — the largest orthopaedic conference in Japan. Dr. Oike will present the lab’s mouse model of periprosthetic joint infection treated with biofilm-targeted monoclonal antibody therapy on May 21, 2026, in Kobe, Japan.
Orthopaedic Research Society (ORS) New Investigator Recognition Award Finalist
Congratulations to Jun Oike, MD, PhD for being selected as an ORS New Investigator Recognition Award Finalist for his work on biofilm-dismantling monoclonal antibody treatment in a mouse model of periprosthetic joint infection.
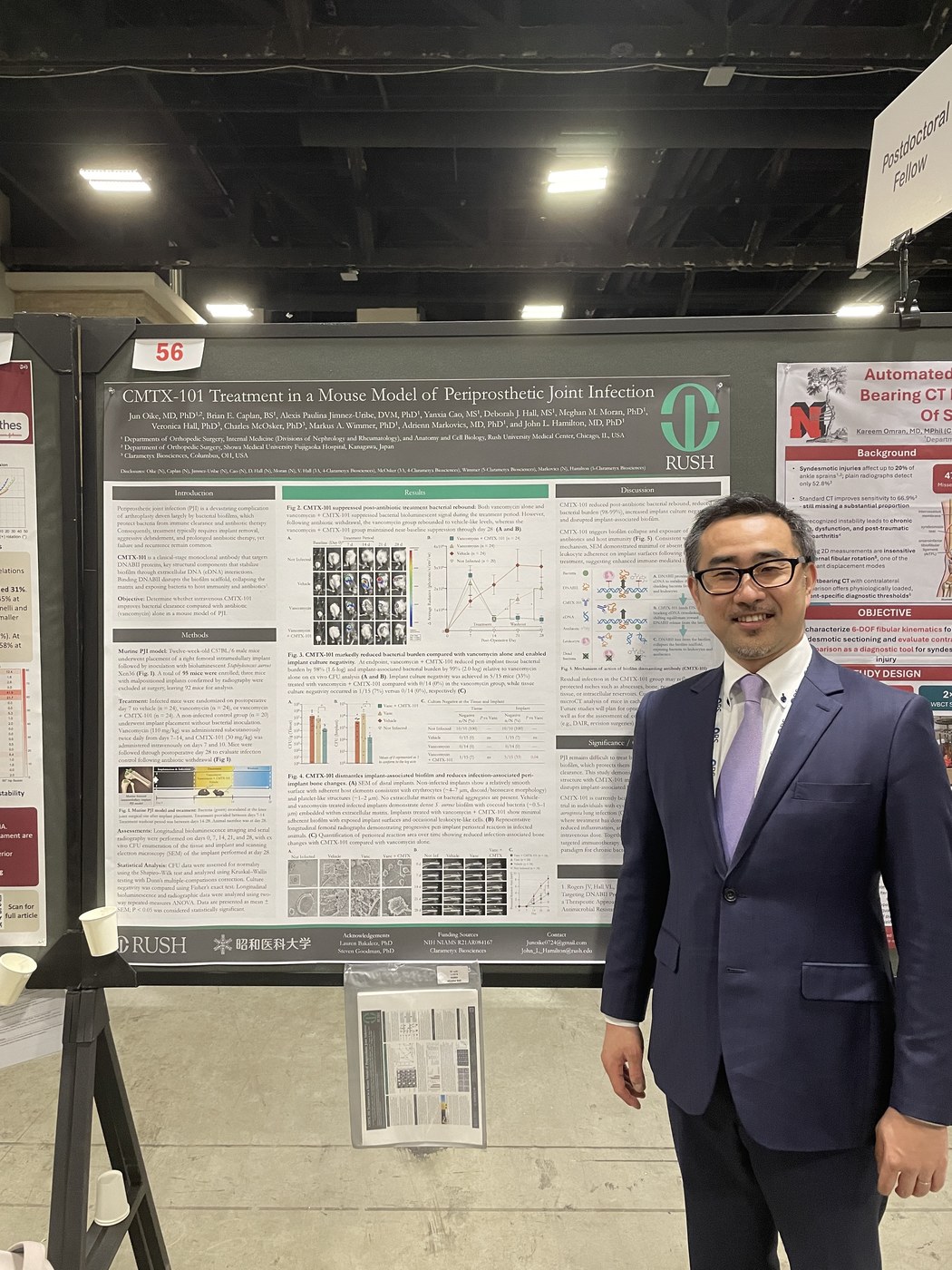
ORS 2026 Annual Meeting, Charlotte, NC.

ORS 2026 Annual Meeting, Charlotte, NC.
ORS Research Interest Group Session: What Will It Take to Defeat Biofilms?
John Hamilton, MD, PhD, Adrienn Markovics, MD, PhD, and Lauren Priddy, PhD, organized and led the Musculoskeletal Infection Research Interest Group session, “What Will It Take to Defeat Biofilms? Translating Scientific Insight to Clinical Impact,” at the 2026 ORS Annual Meeting in Charlotte, NC. John Hamilton, MD, PhD also delivered a separate presentation on emerging anti-biofilm strategies being tested in the clinic.

Jun Oike, MD, PhD accepts the Oral Presentation Award at the 2026 Rush University Trainee Research Day.
2026 Rush University Trainee Research Day Oral Presentation Award: Musculoskeletal Research and Orthopedics
Congratulations to Jun Oike, MD, PhD for winning the Oral Presentation Award at the 2026 Rush University Trainee Research Day for his work on biofilm-dismantling monoclonal antibody treatment and bacterial clearance in a mouse model of periprosthetic joint infection.

John Hamilton, MD, PhD.
NIH/NIAMS Grant Awarded
The MSKI Laboratory was awarded an NIH/NIAMS grant to investigate the use of bacterial biofilm-disrupting antibodies to prevent and treat periprosthetic joint infection.
View on NIH RePORTER
Adrienn Markovics, MD, PhD.
John G. Searle Innovator Award
Congratulations to Adrienn Markovics, MD, PhD on receiving the John G. Searle Innovator Award for the second consecutive year. Dr. Markovics was awarded pilot funding for her project “Targeting Periprosthetic Joint Infection via the Application of mRNA-encoded Lysostaphin.”

Pediatrics Nationwide, Fall/Winter 2024.
Featured in Pediatrics Nationwide Cover Story
The cover story of Pediatrics Nationwide profiles the development of anti-DNABII monoclonal antibody therapy for biofilm-mediated infections. The feature discusses the MSKI Lab collaboration with Lauren Bakaletz, PhD and Steven Goodman, PhD at Nationwide Children’s Hospital and includes commentary from John Hamilton, MD, PhD on the therapeutic potential of anti-DNABII antibodies for periprosthetic joint infection.
Read the full article
Nature Portfolio, Partner Content.
Featured in Nature Portfolio: Biofilm Battles
A Nature Portfolio feature article profiling the anti-DNABII biofilm-targeting therapeutic platform developed at Nationwide Children’s Hospital. The piece discusses the MSKI Lab’s ongoing collaboration with Lauren Bakaletz, PhD and Steven Goodman, PhD and includes commentary from John Hamilton, MD, PhD on the application of anti-DNABII monoclonal antibody therapy to periprosthetic joint infection.
Read the full article
ORS NIRA Finalist & OREF Travel Grant
John Hamilton, MD, PhD was selected as a New Investigator Recognition Award (NIRA) Finalist and received the ORS/OREF Travel Grant in Orthopaedic Research Translation at the 2024 ORS Annual Meeting in Long Beach, California.

Members of the Musculoskeletal Infection Research Interest Group.
Musculoskeletal Infection Research Interest Group
Organized by Adrienn Markovics, MD, PhD and John Hamilton, MD, PhD, the Musculoskeletal Infection Research Interest Group brings together scientists, orthopedic surgeons, infectious disease physicians, fellows, and students at Rush and across the country to discuss the latest research and lab developments in musculoskeletal infection research.
Inquire about the interest group




