Our Approach
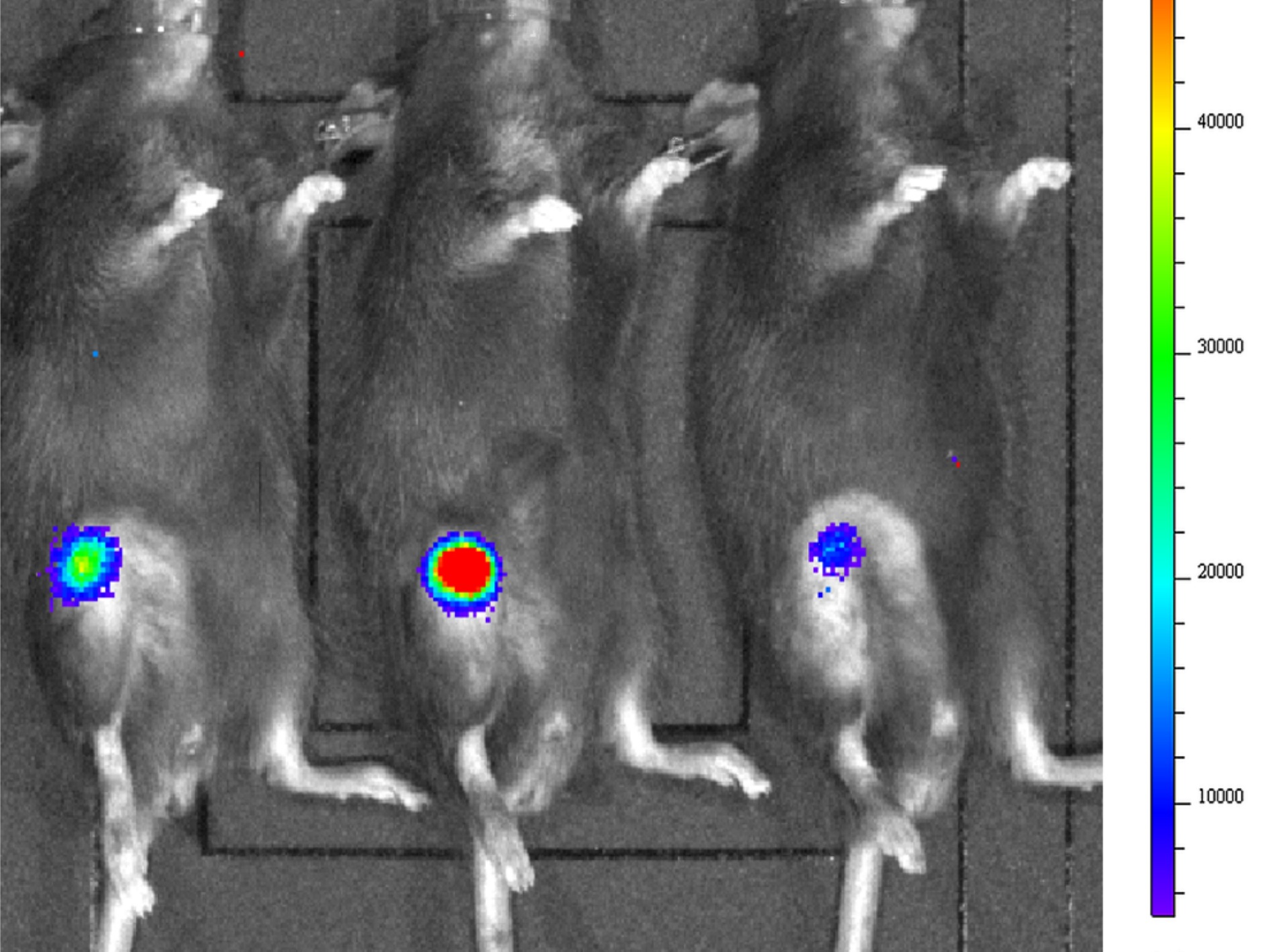
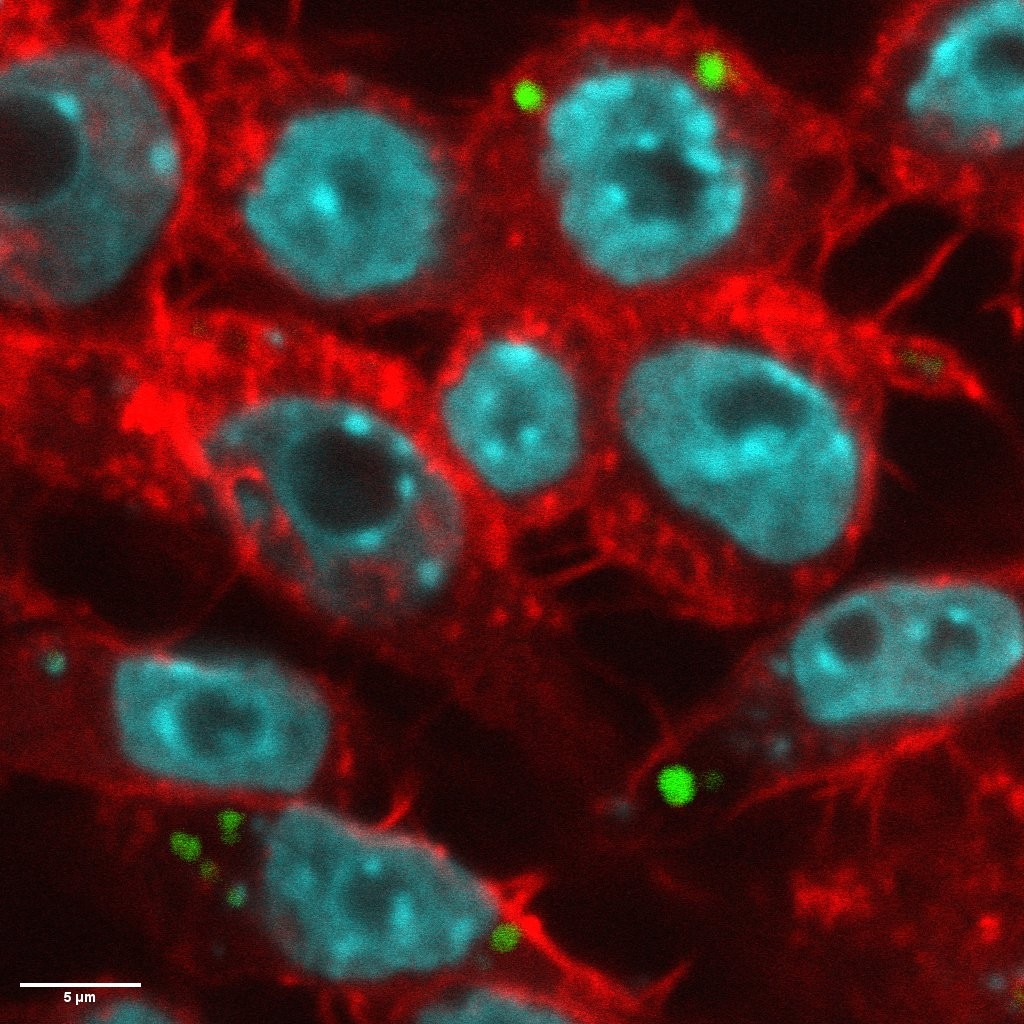
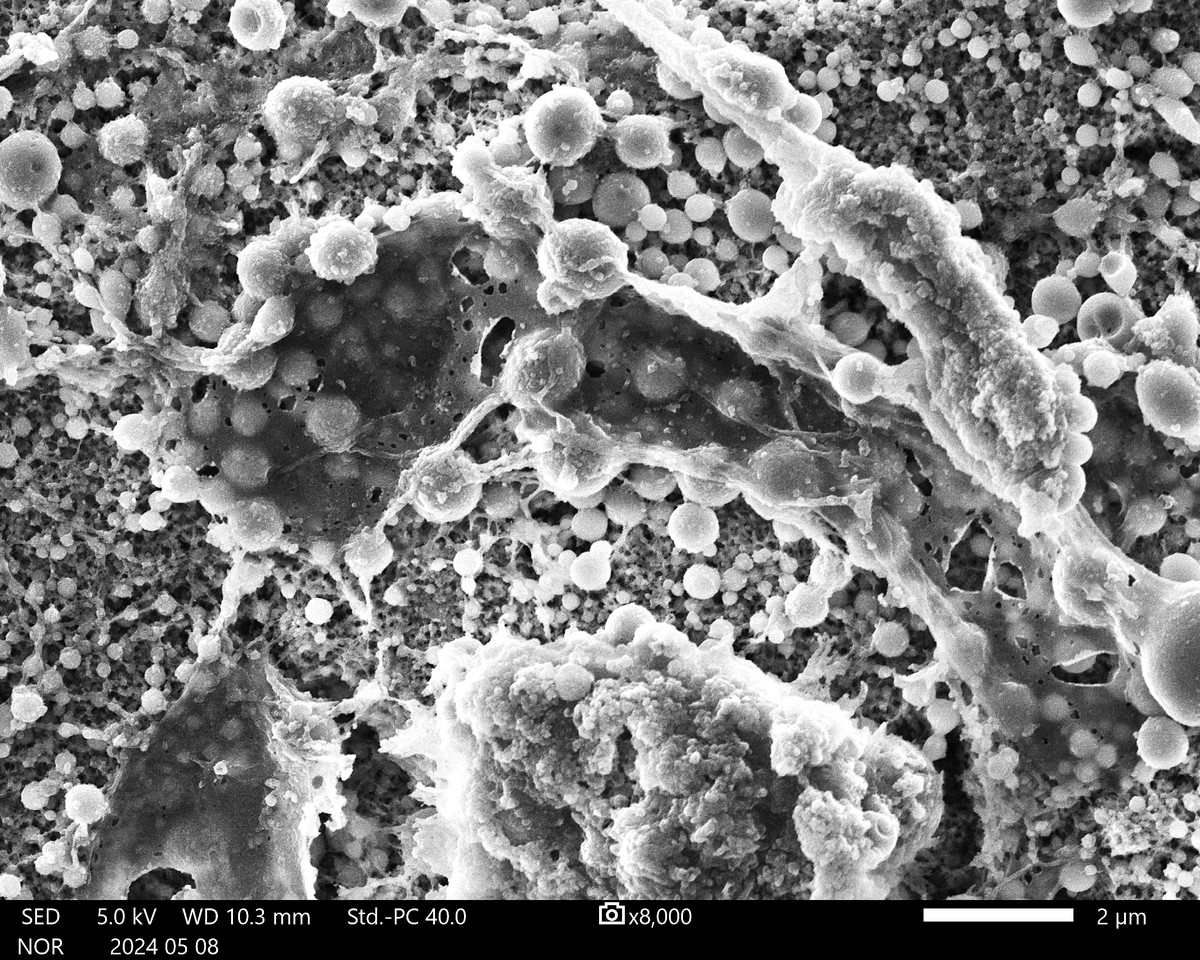
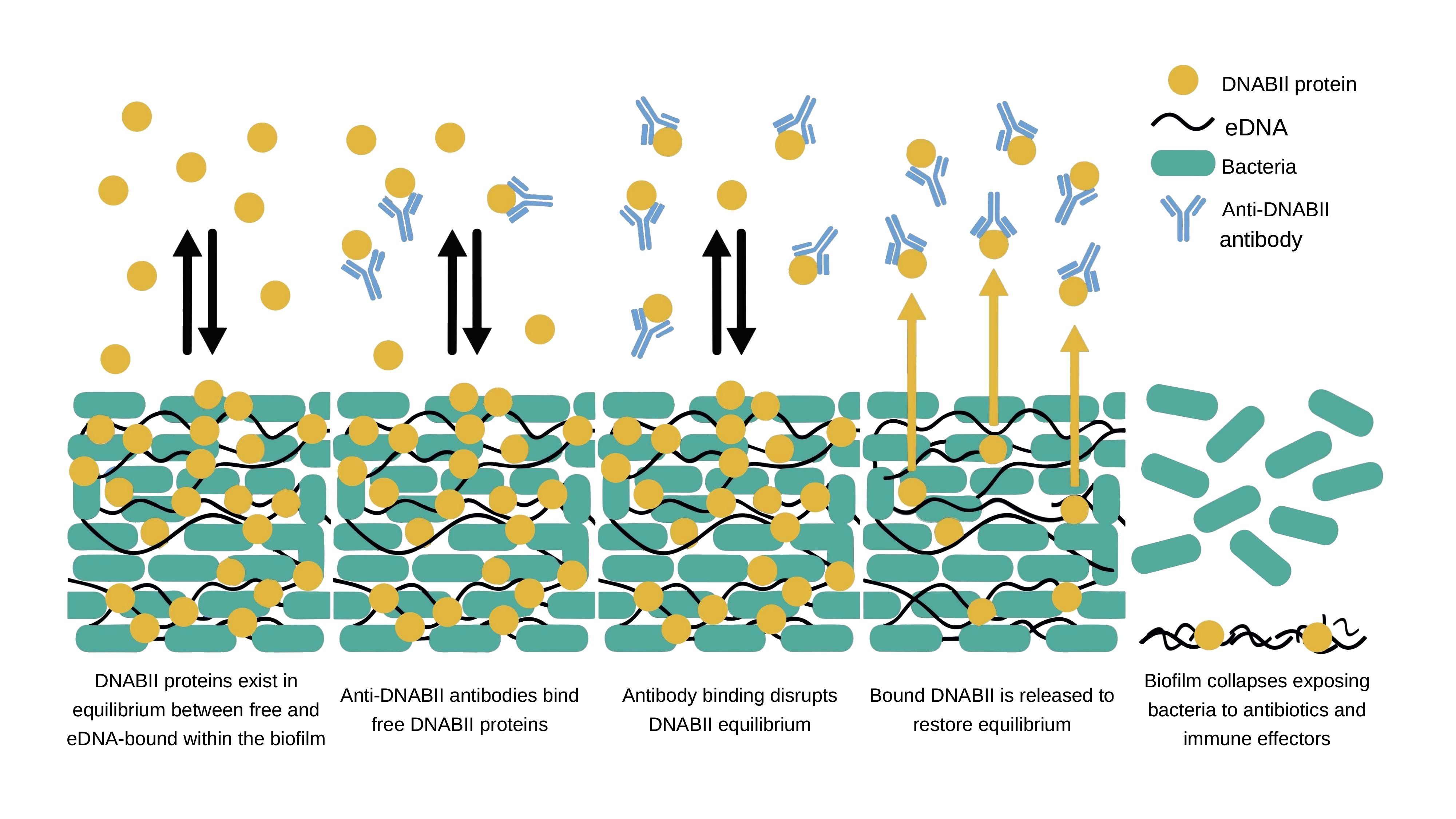
Musculoskeletal infection (MSKI) — including implant-associated infection, such as periprosthetic joint infection and fracture-related infection — remains one of the most challenging complications in orthopedic surgery. Treatment failure is driven by bacterial persistence across protected niches: biofilm, immune-shielded abscess communities, and bacteria residing within bone and host cells. These compartments evade conventional antibiotics and are the central barrier to effective treatment. The MSKI Laboratory integrates molecular biology, immunology, preclinical models, and advanced imaging to define mechanisms of persistence and develop targeted therapeutics.